A $34 Smartphone Gadget That Can Detect HIV in 15 Minutes
Over the years, mobile phones have rapidly progressed from bulky, glorified walkie-talkies to a device capable of performing nearly all the functions of a desktop computer. In today’s world, the ubiquitous smartphone has become an incredibly versatile tool for everyone, including scientists and engineers.
One recent invention proves just how advanced and life-altering this technology has become.

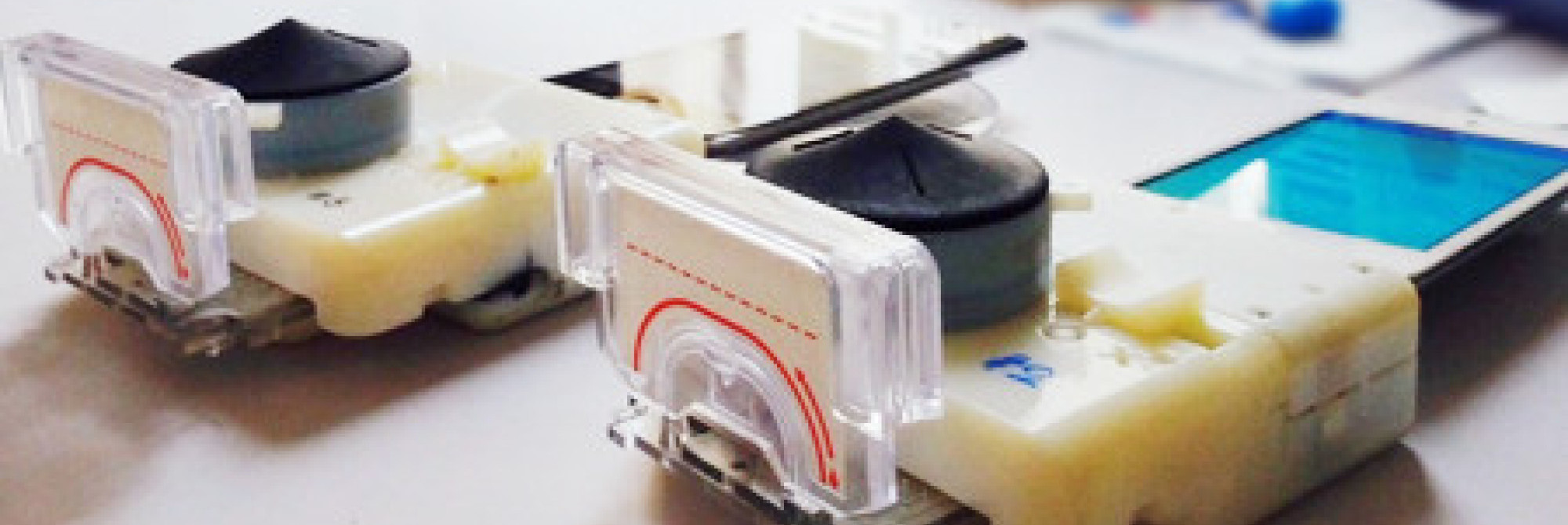
Columbia University biomedical engineering researchers Tiffany Guo and Tassaneewan Laksanasopin have created a device capable of detecting HIV and syphilis in 15 minutes – and its diagnostic powers rely upon a smartphone! The device attaches to any iPhone or Android device through the audio jack and, with one finger prick, a patient drops a small sample of blood onto a cassette that contains a microfluidic chip. That cassette is then inserted into the device and, after just 15 minutes, the results of the test are displayed on the smartphone’s screen.
It does all of this, yet costs only $34 dollars to manufacture.
The affordable gadget was tested during a small clinical trial in Kigali, Rwanda. During that time, 96 patients participated and researchers concluded that the invention is on par with the most accurate HIV diagnostic tests on today’s market. The implementation of such a device in third-world countries where HIV spreads rapidly could save thousands. This is especially apt in the cases of pregnant women who, as the Center for Disease Control (CDC) reports, can lower the risk of passing the disease onto their offspring to under one percent if the HIV is detected early and they are able to begin antiviral medication.
The idea was first conceived in 2007, but took off in 2013 when researchers thought it would be advantageous to harvest the power, portability and affordability of smartphones. “We saw that the smartphone as this ubiquitous device that already had a lot of the components that we wanted,” Guo said. “So we stripped our dongle down to the essentials of what we needed for our assay—very simple optics and very simple fluid control.”
The next step for the device is to seek approval from the World Health Organization, which would allow it to be used to help diagnose thousands of patients in needy countries all over the world.
